|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
This page links to information about the ecology of the Thousand Islands. You can begin below with the Depositional History and Anthropogenic Impacts, or Here to learn about non-native species and how they threaten the ecology Thousand Islands.
|
|
|
Depositional History
The Thousand Islands landform is probably the result of a flood tide delta deposit. Two lines of evidence support this conclusion. Visual evidence from aerial photographs points to a similarity in morphology between the Thousand Islands and known flood tide delta deposits. Additionally, physical evidence from sediment cores indicates the presence of a sand layer consistent with a flood tide delta deposit. Though many of these islands have been modified by dredging, the Thousand Islands formation is natural.
In Figure 1, aerial views of the Thousand Islands prior to significant modification and a recently deposited flood tide delta in North Carolina are compared. Similarities in general morphology can be seen. Long-shore transport has since closed the inlet at Cocoa Beach, though a topographical low area still exists where the inlet probably once was.
|
|
|
|
 |
|
|
|
Figure 1. The Thousand Islands (left) in 1956, before significant dredging, compared to a flood tide delta behind a barrier island in North Carolina. Cocoa Beach photo courtesy Brevard County Mosquito Control. Flood tide delta picture from Barrier Islands S.P. Leatherman Ed., page 216.
|
|
|
|
Lasater and Nevin (1973) took several shallow cores in the Thousand Islands as part of a report to a property owner. Though some of their sampling locations were close to dredging sites and their analysis was limited, the cores did exhibit characteristics consistent with a flood tide delta deposit. This is principally the existence of a layer of sand, free of the shells that are indicative of deposition in a normal benthic environment (Cooper, 1994; Armon, 1979; Moslow and Heron, 1979). The present author has also done some shallow core work (<1m) in the islands south of Minutemen Causeway, finding the same general trends. Shells representing a benthic community are present in the upper approximately 10cm of sediment. Below this is a layer of varying thickness, composed of sand, sometimes interspersed with pieces of woody vegetation. Below the sand layer, the cores generally exhibit the shell sand/silt layer similar to the benthic community found at the core surface. This uneven sand layer lacking shells suggests an interruption in the normal accretion of benthic sediments by a rapid deposition of material being washed in by wave action. The date of this event is unknown.
Anthropogenic Impacts
Aboriginal people inhabited Brevard County for centuries before the arrival of Europeans (Cultural Resource Management, 1978). Shell middens are present in at least two areas (see appendix 1 for locations) of the islands and the author has found pottery sherds along the barrier shore prior to the construction of River Lakes condominium near 8th Street south.
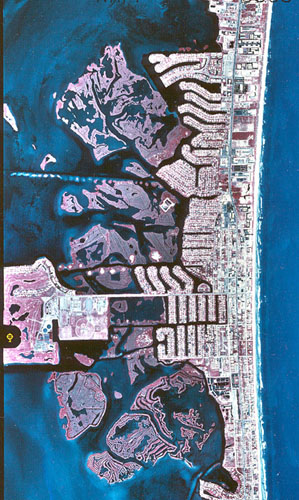
Since World War II, the Thousand Islands have been heavily impacted by development and mosquito control (Figure 2). Beginning in the late 1950s, small ditches were dug through the islands to facilitate fish access to inner areas of the Sarcocornia marsh for mosquito control (Scott Taylor, personal communication). At approximately the same time, dredge and fill activities were begun in Cocoa Beach for housing development. This involved dredging of canals to provide fill material for houses. During the late 1960s deeper ditching by dragline was used as a stronger effort to control mosquitoes. The islands south of Minutemen Causeway were more heavily impacted in this way than those islands to the north. In 1972 the impoundment known as C-34 was begun on 100 acres of state-owned islands near the high school. This section of islands was completely impounded with a perimeter dike to facilitate mechanical flooding of the marsh during mosquito breeding season. This impoundment is no longer actively managed in this way (Scott Taylor, personal communication).
|
|
|
|
 |
|
|
|
Figure 2. Aerial photograph of Thousand Islands and Cocoa Beach, 1994.
|
|
|
|
Effects on Habitat
Housing development and mosquito control have had several effects on the habitat within the Thousand Islands. The construction of Minutemen Causeway has severely disrupted water movement through the islands, particularly those to the south of the road. This impedes flushing and solute exchange between the wetlands within the islands, and the Banana River Lagoon. To eliminate mosquito breeding habitat, the naturally shallow angle of repose of the shore/water interface has been greatly increased. This affects hydrologyand interferes with several of the ecosystem processes that normally take place at this interface. Soils have been raised above the water table, allowing upland plant species to move in, changing the species composition of the islands. Paradoxically, this has increased the species diversity of vascular plants there. Construction of dikes has apparently decreased the breeding habitat of the horseshoe crab (Limulus polyphemus), and the diamondback terrapin (Malaclemys
terrapin) and has probably facilitated easier access for raccoons to nests, increasing predation on turtle eggs. Despite the grim prediction of Lasater and Nevin (1973), the Thousand Islands have not become "ecologically dead". The area supports large numbers of migratory birds, native wading birds, ospreys, alligators, small mammals, and even an occasional otter is seen there.
One effect of mosquito control dredging in the southern portion of the Thousand Islands was the creation of a maze of small canals that are useful for canoeing (Figure 3). The added depth in these small canals has greatly decreased the amount of light that is available to submerged aquatic vegetation, effectively eliminating it from these areas. Consequently, the main source of primary productivity within these canals is planktonic algae. The water is often very low in dissolved oxygen (< 1ppm) below a depth of one meter (Kozusko, 1995, unpublished data). Fish-kills from oxygen stress appear to be rare, however.
|
|
|
|
 |
|
|
|
Figure 3. Aerial view of a section of Thousand Islands showing canals. Photo by T. Kozusko, July, 1999.
|
|
|
|
The dredging of channels for housing development has created several islands with substantial areas of upland habitat (see Appendix 1 for locations). These areas could be used for passive recreation. Hiking trails, interpretive signs, and an open picnic area are possibilities. They have been used for interpretive trails for field trips for elementary students (see Kayaks and Compasses).
Vegetation Communities
The vegetation communities of the Thousand Islands can be classified in three habitat types: natural marsh, dredge-spoil, and tropical hammock associated with shell middens. Of these three, the dredge-spoil community contains the highest plant diversity and suffers the greatest degree of invasion by non-native plant species.
The native marsh habitat is found inside the C-34 impoundment and along most of the edges of the islands. The margins of the islands and their interiors along ditches are typically fringed with mangroves (Figure 4). Unaltered islands have mangroves inland if they are inundated sufficiently to allow propagule dispersal to the interior. The dominant plants of the unaltered interior marshes appear to be Sarcocornia perennis and Batis maritima.
|
|
|
|
 |
|
|
|
Figure 4. Aerial view of Sarcocornia and Batis marsh with mangroves established along mosquito control ditches. Photo by T. Kozusko, July, 1999.
|
|
|
|
In Brevard County mangroves are near the northern limits of their ranges and suffer mortality and disturbance from freezes. When mangroves die back, Sarcocornia and Batis are allowed to expand. These plants will persist unless mangroves become established and shade them
out. This typically takes place until the next freeze kills the mangroves back. There are several areas where saltmarsh cordgrass (Spartina alterniflora) maintains dense stands that have persisted for several decades. This species is well known in northern salt marshes and begins to dominate in northern Brevard County. Blackrush (Juncus roemerianus) is not found in the Thousand Islands. Saltgrass (Distichlis spicata) exhibits only limited coverage.
Dredge spoil harbors the most diverse vegetation community in the Thousand Islands (see Appendix 2). The change in soil regime from wetland to upland resulting from spoil deposits allows disturbance invaders to colonize areas previously tolerated only by species with adaptations to water-logged saline soils. The major non-native species found in this habitat are Brazilian pepper (Shinus terebinthifolius) and Australian pine (Casuarina glauca, and C. equisetifolia.). However, the native species swamp privet (Forestiera segregata) and salt bush (Baccharis halimifolia) probably exhibit the greatest coverage. An aerial view of typical dredge spoil habitat is given in Figure 5 and a ground-level view is presented in Figure 6.
|
|
|
|
 |
|
|
|
Figure 5. An aerial view of typical dredge spoil habitat. Much of the vegetation
visible in this view consists of Schinus terebinthifolius, Forestiera segregata, and Baccharis halimifolia. Photo by T. Kozusko, July, 1999.
|
|
|
|
 |
|
|
|
Figure 6. Ground-level view of typical dredge spoil habitat. Plants visible are Forestiera segregata, Baccharis halimifolia, and in the foreground are the cactus Opuntia stricta. The dark green in the upper left and at right is Conocarpus erectus (buttonwood). Photo by T. Kozusko, October, 2000.
|
|
|
|
Tropical vegetation is found in several locations in the southern Thousand Islands (Appendix 1, Appendix 4). The two major areas are both associated with shell middens. The small area north east of C-34 consists of a few trees that have volunteered in dredge spoil.
Tropical genera present on the C-34 hammock before dike work included Amyris, Bursera, Capparis, Chiococca, Erythrina, Eugenia, Ficus, Randia, and Torrubia (Bidlingmayer, unpublished vegetation survey, 1971). Many of these species are listed by Wunderlin (1998) as having the northern limit of their range in or near Brevard County.
In 1999 (unpublished report) the vegetation of the Thousand Islands was listed by this author, and the C-34 hammock area was photographed by him from the air in July of the same year (Figure 7). All the species found by Bidlingmayer (1971, unpublished) were found. Another tropical hammock associated with a shell midden was discovered by this author on an island southeast of C-34 (Appendix 1). In a recent preliminary survey for a research proposal, several of the tropical species were noted on the perimeter dike near the shell midden.
|
|
|
|
 |
|
|
|
Figure 7. Aerial view of C-34 hammock area, looking north, July, 1999. Photo by Tim Kozusko.
|
|
|
|
Several factors might account for the association of tropical species with shell middens. Two likely factors are the increased soil aeration, and the ability of calcium carbonate from shells to ameliorate soil acidity usually associated with oxidation of the sulfides created by anaerobic metabolism (Norman, 1976). Additionally, the volume of water in the Banana River acts as a thermal mass, mitigating detrimental effects from cold fronts. Dredge spoil material also contains shells and might be chemically similar to soils of shell middens. Some tropical species have volunteered at a site approximately 500m from the nearest seed source (Appendix 1). In the C-34 impoundment the tropical hammocks have spread out onto the perimeter dike. Several species have dispersed to the south of the eastern hammock (Appendix 4). It seems likely that this plant association might be induced to colonize the areas of upland habitat in the Thousand Islands.
Literature Cited and Appendices Here.
Acknowledgments:
Dr. W.K. Taylor, Don Spence, and Kate Buchanan provided assistance with tropical plant identification. Scott Taylor made many documents available from Brevard County Mosquito Control.
|
|
|
|